Globalization, criminal activity, lax regulation and traceability requirements, and less-than-stringent production protocols have increased the incidence of potentially dangerous counterfeit medication and medical devices in the marketplace. It is estimated that 10% of all the medication sold worldwide is actually counterfeit—useless placebos at best, and dangerous substances at worst. This poses a risk to consumers, patients and manufacturers. Serialization is an effort to make tracking and verifying medications and devices faster, easier and more accurate.
Food products, both for humans and for animals, are coming under tighter regulatory compliance by way of the Food Safety Modernization Act (FSMA). Food, like medicine, has become very globalized and very complex, and contains some of the consumer dangers inherent in medicine with issues like allergic reactions.
Pharmaceutical
Global pharmaceutical companies lose an estimated $75 billion annually to counterfeit, gray market and stolen product. Impending regulations aimed at protecting public health, intellectual property and national security will require pharmaceutical, medical device and consumer packaged-goods manufacturers to track and trace products across the supply chain.
With regards to medication specifically, in response to weaknesses in the regulation of the drug supply chain, the US Congress passed the Drug Quality and Security Act on November 21, 2013. Title II of the Act, the Drug Supply Chain Security Act, requires that manufacturers affix or imprint a product identifier to each package and homogeneous case (pharmaceutical serialization). Tracing your finished product back to API’s, compounds, and all upstream suppliers is critical. And tracking downstream is equally important in the event of a recall.
Medical Devices
The FDA requires manufacturers to comply with the final rule establishing a unique device identifier (UDI) released in 2013. This rule was designed to adequately identify medical devices from manufacture throughout it’s useful life using a UDI as issued under a system operated by an FDA-accredited issuing agency. The UDI is to consist of two parts: a) a device identifier that identifies the manufacturer and its specific version or model and (b) a product identifier that identifies one or more of the following:
- Lot or batch number
- Device serial number
- Expiration date (if applicable)
- Manufacture date
- The distinct identification code required by §1271.290(c) for a human cell, tissue, or cellular and tissue-based product (HCT/P) regulated as a device.
Food and Beverage
In the food and beverage vertical, manufacturers must implement the FSMA which also increases requirements for traceability not only of finished product but also of ingredients, batches, and potential allergens. Traceability of batches upstream to supplier lots and downstream to finished good SKU’s and shipping pallets is critical. Allergen verification, CIP’s and packaging material are all critical to your operations. Speed of data and granularity of data increase both the consumers’ protection and your company’s protection.
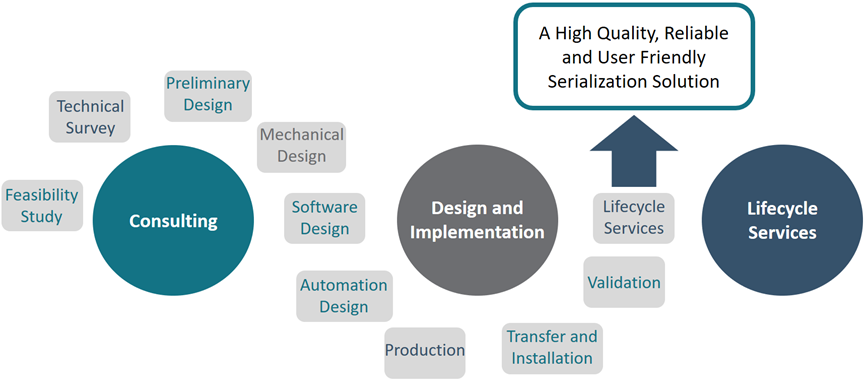
How Grantek can help
Because Grantek has extensive experience in the pharmaceutical and medical devices industries as well as the food and beverage industry, combined with our experience with FDA and Health Canada regulations, we have the unique capability to help our customers successfully and safely comply with these new regulations. We are equipped to help by integrating technologies and processes to allow our clients to implement serialization of products coming off of the production line, as well as provide enhanced traceability for ingredients.
Within the four walls of the factory, implementing the new regulations requires tracking at every stage in the end-of-line packaging process down to machines, printers, labelers, bar-code scanners and vision systems. At each stage in the finished goods packaging process, a unique serial number – represented by a 2-D barcode or RFID tag – must be applied to the product’s package. Depending on the industry or customer’s need, there can be up to five levels of parent/child aggregation, with serial numbers and other batch or product information applied to the item level or unit dose, up to the pallet. The unique serial number at each stage in the packaging process must be married to all the unique serial numbers contained in the package. Therefore, each pallet serial number (parent) is paired to all the unique case serial numbers (children) on the pallet, creating the aggregated parent/child relationship. Likewise, each case serial number (parent) is paired to all of the unique carton or bottle or bundle or other unit serial numbers (children) that are in the case, and so on. Accurately applying and associating these new and complicated sets of individual identifiers poses interoperation challenges for many manufactures.
Grantek utilizes a holistic approach to design and utilizes off-the-shelve components in their delivered systems. This allows for long-term support and easy ordering. Grantek works with industry leaders and suppliers of globally based technologies to ensure the hardware supply chain is not limited to Grantek.
We can provide thought leadership, in front-end design, development, implementation, commissioning, testing, and qualification of MES, LIMS, Historians, vision systems, barcode reader applications, and on-the-line integrated printer applications, as well as convert data output from these systems into comprehensive reports to help manufacturers track ingredients and components as well as finished product, and maintain serialization data for regulatory compliance.
